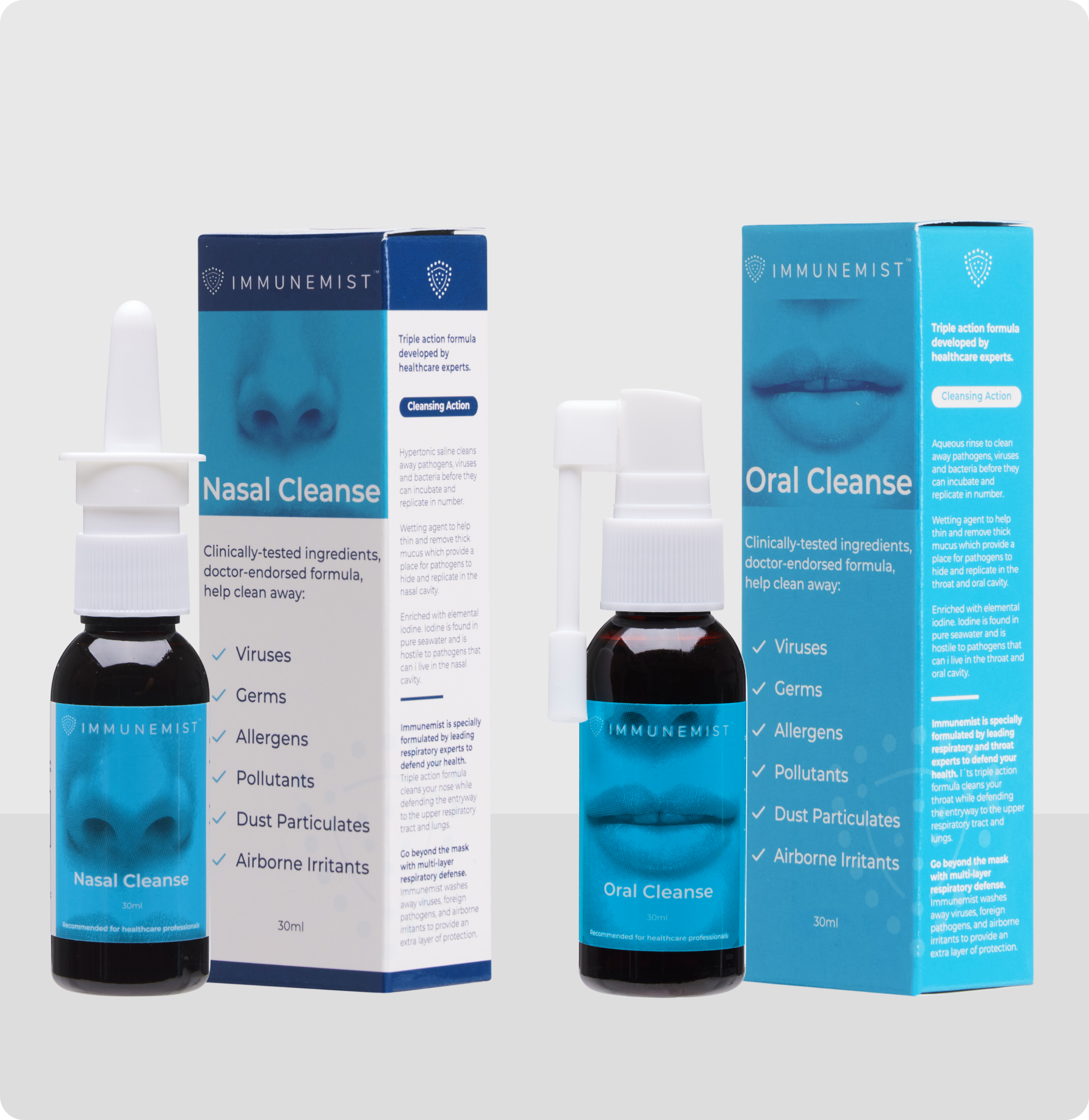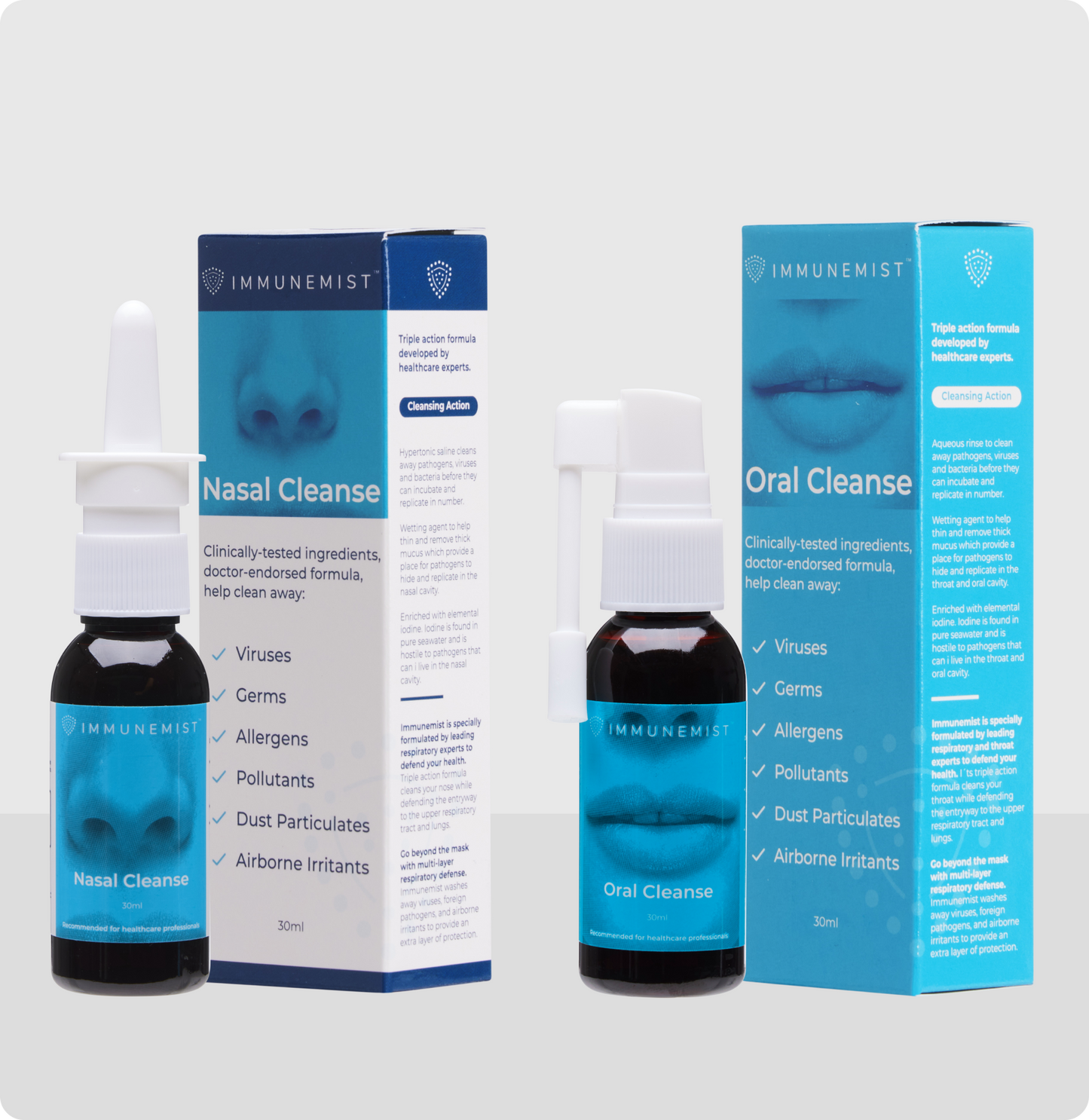

The Power of IMMUNEMIST®
Why ImmuneMist®
& Who is it for?
Inhaled virus particles and bacteria can stay suspended in the air. Once inhaled they then deposit in the upper respiratory tract where they incubate. Most studies suggest it takes 3-7 days after initial exposure for viral load to replicate to the level of contracted infection. Studies throughout the world prove that nasal and oral disinfection and cleaning are key to preventing a virus from establishing a foothold in the body. The ImmuneMist® Nasal and Oral Cleanse is a comprehensive system that keeps your upper respiratory tract clear of pathogens, and reduces viral load in the event of exposure, ImmuneMist® is an effective added layer of protection recommended by leading Doctors and Pharmacists around the globe. This product may be recommended to the general public, and is especially relevant for: essential workers, business travelers, those who engage in social gathering or public congregation, general crowd exposure, offices and classrooms, and in high transient environments such as airports, hotels and public transportation.
Directions for Use
-
Step 1
Insert Into Nostrils
Keep head upright and insert tip of nasal spray pump into your nostrils approximately 1/4 inches.
-
Step 2
Spray Into Nostrils
Using two fingers on the pump with your palm or thumb supporting the base of the bottle, pump between one and tree (1-3) full sprays into each nostril. Sniff gently to then draw solution into nasal cavity.
-
Step 1
Spray Back of Throat
Keep head upright and insert spray tip partly into the mouth, spray 2-3 sprays toward the back of the mouth (throat area).
-
Step 2
Wait 15 Seconds
Waiting fifteen seconds, then expectorate (spit out) the remaining liquid into a sink or clean tissue. Avoid swallowing spray.
What is ImmuneMist® and how does it shield you from inhaled particles and pathogens?
ImmuneMist® is available over-the-counter. Its nasal and oral cleansing system provides three benefits to you:
- Its superior formulas help remove inhaled foreign particles, viruses, germs and stagnant mucus from your nasal and oral cavities. This is also called nasopharyngeal cleansing, a practice continuing to be highlighted in importance by the world’s leading experts in preventative care.
- Its portable and “easy on the go” format makes it convenient to use day to day, while traveling, and after instances of public exposure.
- The ImmuneMist® System (Nasal + Oral Cleanse) provides superior cleansing to that of conventional nasal sprays. It is well known that inhaled particles come into the nose and exit the mouth during coughing. Therefore, it’s imperative to decontaminate both the nasal and oral cavities.
Who is ImmuneMist® for?
ImmuneMist® is safe for anyone ages twelve (12) and older. ImmuneMist® is especially relevant for: healthcare and essential workers, high-risk individuals, after instances of indoor public exposure, after events and social gatherings, for those who spend time in communal spaces such as offices and classrooms, as well as for travelers with exposure to hotels, airports, and/or other formats of public transportation.
Is ImmuneMist® safe?
Yes. 100% of the ingredients have been used safely for many decades. All ingredients are recognized as safe by FDA and international health agencies.
Is ImmuneMist® Regulated by FDA?
ImmuneMist® is regulated under the Food, Drug, and Cosmetic Act and is produced in an FDA approved facility in the USA. ImmuneMist® is a cleansing product.
Does ImmuneMist® shield you from respiratory threats?
Yes, the ImmuneMist® cleansing action works effectively against respiratory threats and to clean-away germs and viruses.
Where is ImmuneMist® produced?
ImmuneMist® is proudly produced in the USA in an FDA approved facility.
Why are the “Ask your doctor if you have…” warning conditions on the box?
Each person’s individual medical history is only known to that person and their doctor. This is why you should discuss these and any warning with you doctor if they apply to you, such as thyroid disease and allergy to any of the ingredients.
What ingredients are in the ImmuneMist® formulas?
Nasal Cleanse Ingredients: Povidone Iodine, Citric Acid Monohydrate, Sodium Citrate Dihydrate, Sodium Chloride, Glycerine, WS-23 (cooling agent), Raspberry Flavor, Purified Water. ImmuneMist® Nasal Cleanse is a tea-colored liquid.
Oral Cleanse Ingredients: Povidone Iodine, Citric Acid Monohydrate, Sodium Citrate Dihydrate, Sucralose, Potassium Lodine, Glycerine, WS-2 (Cooling Agent), Raspberry Flavor, Purified Water. ImmuneMist® Oral Cleanse is a tea-colored liquid.
Is ImmuneMist® Doctor & Pharmacist Recommended?
Yes, ImmuneMist® is recommended by Doctors, Nurses, Nurse Practitioners and Pharmacists. It is recommended by numerous organizations that are leading early treatment education and efforts for better public health during these challenging times. ImmuneMist® was developed by leading healthcare experts. They bring more than 80 years of experience in developing consumer and medicinal products in the USA and abroad.